Snee Group reports record-breaking quantum dot reducing agent
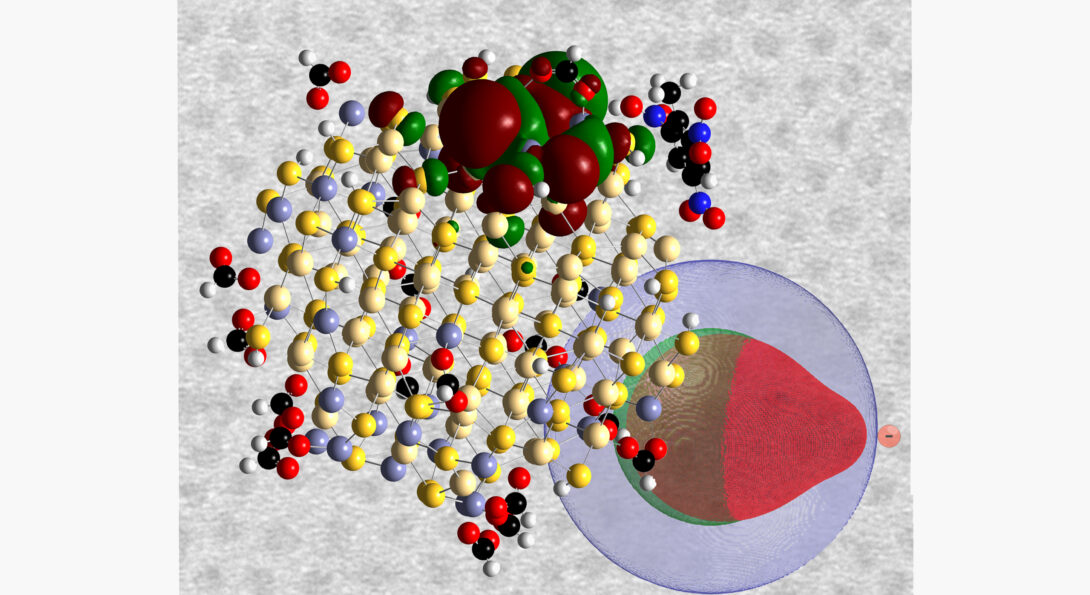
The Snee group recently reported a highly efficient quantum dot photoreducing agent in the journal Nanoscale. Quantum dots have been studied for their size-tunable electronic and optical properties, and recently they have been used as redox agents. This allows them to generate solar fuels from sunlight and reduce CO2. Unfortunately, QDs are unstable because they quickly precipitate in solution, damaging their surfaces and rendering them useless. Overcoating the quantum dots solves the problems with stability; however, conventional wisdom states that the shell prevents the ability of QDs to reduce substrates.
The group was collaborating with researchers from the US Army when they discovered that core-shell CdZnS/ZnS QDs are 300% more efficient than the core QD alone when reducing organic substrates. The reason for the enhanced efficiency was that the ZnS shell prevented trapping of the semiconductor’s electron and hole by surface defects, allowing the quantum dot to remain in the excited state for a longer period of time. As for why the shell doesn't present a barrier for reduction, it was found that the semiconductor hole was able to penetrate into the shell, which allows the electron and hole to remain in close contact. This close pairing minimizes the energy of the reduced substrate, which enhances the reaction rate. The unexpected demonstration of enhanced redox activity of core-shell CdZnS/ZnS will impact on several scientific fronts especially due to the substantial stability of core-shell QDs; several news organizations have highlighted this report.