Weakened antibodies become powerful tools for superresolution imaging
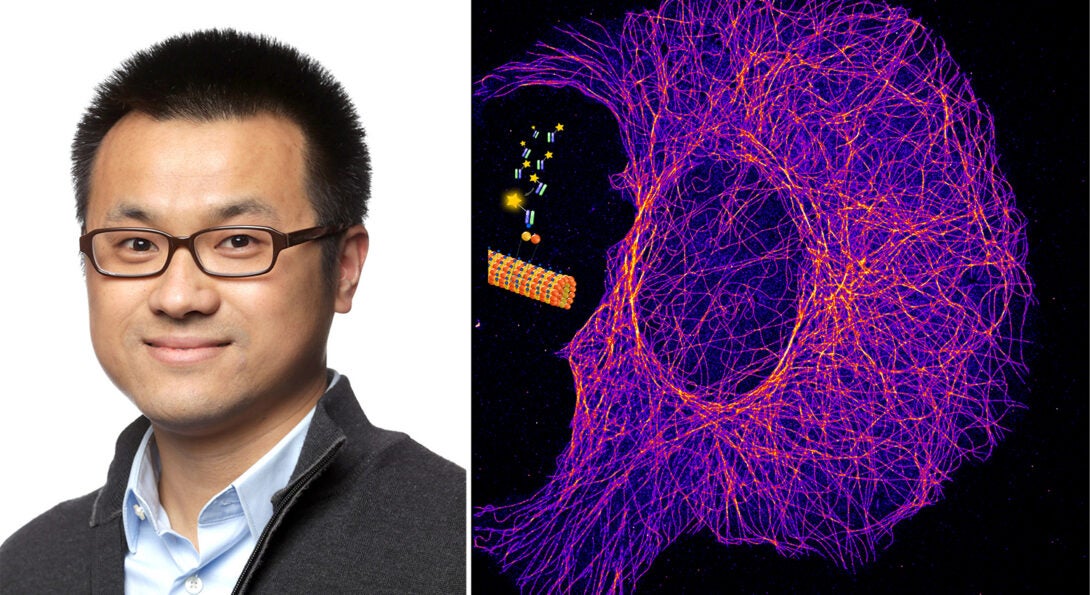
Antibodies with lowered affinities can overcome a major limitation in superresolution imaging, according to new research from the Hu group.
High-affinity antibodies are widely used to label specific protein targets for superresolution imaging. However, with achievable resolution down to the size of a single antibody molecule, the labeling technique creates artifacts that render molecular quantification error-prone.
The research team, led by Ying Hu, has turned to antibodies with loose binding. They discovered that short-lived interactions could overcome labeling artifacts associated with tight binding. By generating fragments of antibodies and applying a chemical reagent that disrupts noncovalent interactions, the group created a new superresolution technique using antibodies against a short peptide tag.
"Antibodies are immune system's scouts," said Hu, assistant professor of chemistry. "They bind tightly to foreign invaders and stop them from harming the body. What's surprising to us is we could lower the binding strength of an antibody and still show its high binding specificity. This was not obvious because, in the literature, high affinity usually infers high specificity, and our data suggest there may be more to the story."
When compared to existing superresolution techniques using standard antibody labeling, they observed a significant improvement in the consistency of molecular quantification.
"The coolest feature is the simplicity and the versatility of the technique," said Hirushi Gunasekara, the lead author of the study and a chemistry graduate student in the group.
“Our ability to precisely eliminate parts of the antibody combined with single-molecule sensitivity also allows us to dissect complex antibody behaviors, like antibody-dependent enhancement of SARS-CoV-2 infections," Hu added.
The work titled "Chaotropic Perturbation of Noncovalent Interactions of the Hemagglutinin Tag Monoclonal Antibody Fragment Enables Superresolution Molecular Census" is published in ACS Nano.