Tom Driver
Professor
Contact
Address:
3220 SELE, MC 111
Office Phone:
Email:
Related Sites:
About Heading link
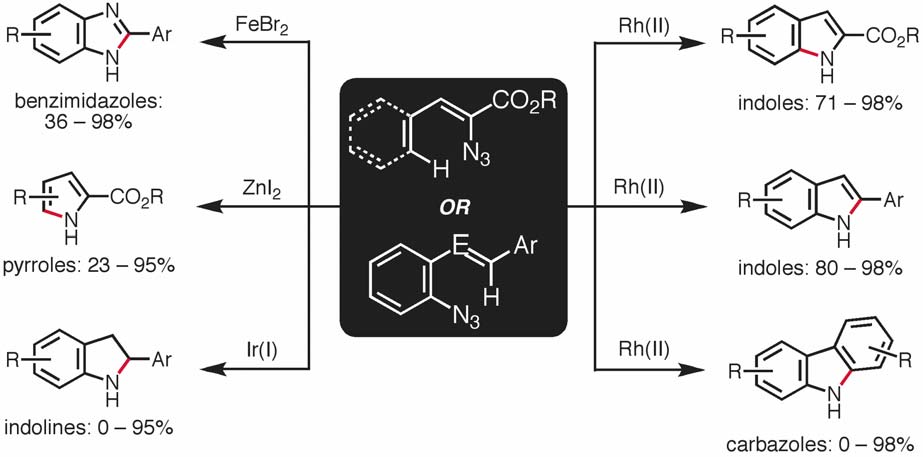
My research group is focused on the rational development of new transition metal-catalyzed methods that selectively transform simple molecules into functionalized N-heterocycles. Towards that end we are interested in (1) exploring the chemistry of azides and (2) functionalizing traditionally unreactive C–H bonds.
We have recently discovered a number of transition metal-catalyzed methods, which enable the conversion of azides to a variety of important N-heterocycles (Scheme 1). We aim to elucidate the mechanism through which these processes operate and develop new methods based on our mechanistic conclusions.
Selected Publications
1. Stokes, B. J.; Richert, K. J.; Driver, T. G., Examination of the Mechanism of Rh2(II)-Catalyzed Carbazole Formation Using Intramolecular Competition Experiments. J. Org. Chem. 2009, 74, 6442.
2. Sun, K.; Sachwani, R.; Richert, K. J.; Driver, T. G., Intramolecular Ir(I)-Catalyzed Benzylic C–H Bond Amination of ortho-Substituted Aryl Azides. Org. Lett. 2009, 11, 3598.
3. Shen, M.; Driver, T. G., Iron(II) Bromide-catalyzed Synthesis of Benzimidazoles from Aryl Azides. Org. Lett. 2008, 10, 3367.
4. Shen, M.; Leslie, B. E.; Driver, T. G., “Dirhodium(II)-catalyzed Intramolecular C–H Amination of Aryl Azides.” Angew. Chem., Int. Ed. 2008, 47, 5056.
5. Dong, H.; Shen, M.; Redford, J. E.; Stokes, B. J.; Pumphrey, A. L.; Driver, T. G. “Transition metal-catalyzed synthesis of pyrroles from dienyl azides.” Org. Lett. 2007, 9, 5191.
6. Stokes, B. J.; Dong, H.; Leslie, B. E.; Pumphrey, A. L.; Driver, T. G. “Intramolecular C–H amination reactions: exploitation of the Rh2(II)-catalyzed decomposition of azidoacrylates.” J. Am. Chem. Soc. 2007, 129, 7500.
Education
- BSc (Honors, Highest Distinction) 1999, Indiana University
- PhD, University of California, Irvine, 2004
- NIH Ruth L. Kirschstein NRSA Postdoctoral Fellow, California Institute of Technology, 2004-2006